Most fluid pharmaceutical products are not simple liquids and do not follow Newton’s law of flow. These fluids are known as non-Newtonian fluids. Non-Newtonian behavior is typically seen in liquid and solid heterogeneous dispersions, such as colloidal solutions, emulsions, liquid suspensions, and ointments. When non-Newtonian materials are analyzed using a rotational viscometer and the results are plotted, various consistency curves emerge, representing three classes of flow: plastic, pseudoplastic, and dilatant.
Plastic Flow
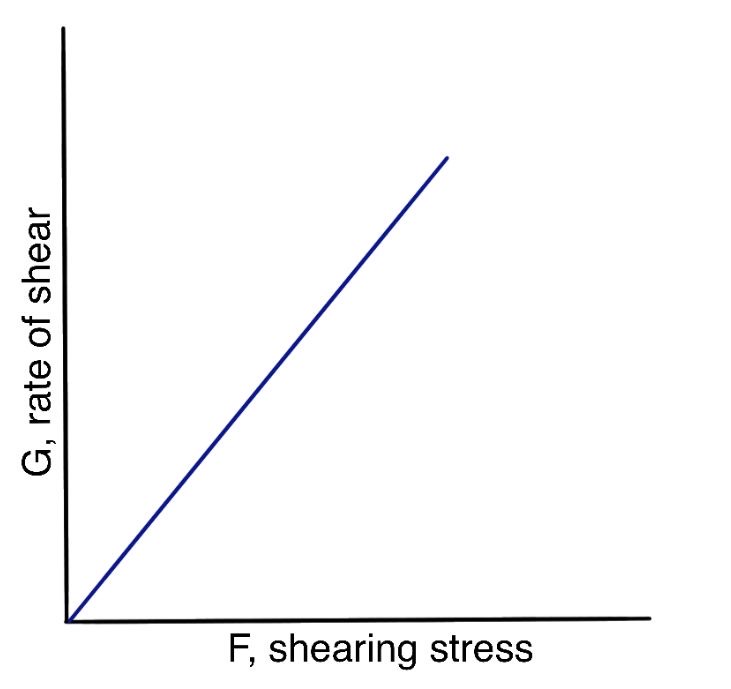
In Figure 1, the curve represents a material that exhibits plastic flow; such materials are known as Bingham bodies, named after the pioneer of modern rheology who first systematically studied plastic substances.

Plastic flow curves do not pass through the origin but intersect the shearing stress axis (or would if the straight part of the curve were extrapolated to the axis) at a specific point called the yield value. A Bingham body does not begin to flow until the shearing stress exceeds this yield value. Below the yield value, the substance behaves as an elastic material. Rheologists classify Bingham bodies, which exhibit a yield value, as solids, whereas substances that begin to flow at the slightest shearing stress and show no yield value are defined as liquids. Yield value is an important property of certain dispersions.
The slope of the rheogram in Figure 1 is termed the mobility, and its reciprocal is known as the plastic viscosity, U. The equation that describes plastic flow is:
$$ U=\frac{F-f}G $$
where f is the yield value, or the intercept on the shear stress axis, measured in dynes/cm², and F and G are defined as before.
Plastic flow is associated with the presence of flocculated particles in concentrated suspensions, resulting in a continuous structure throughout the system. A yield value exists due to the contacts between adjacent particles, caused by van der Waals forces, which must be overcome for flow to occur. Therefore, the yield value indicates the force of flocculation: the more flocculated the suspension, the higher the yield value. Frictional forces between moving particles can also contribute to the yield value. a plastic system behaves like a Newtonian system at shear stresses above the yield value.
Pseudoplastic Flow
Pseudoplastic flow is typically observed in polymer solutions, unlike plastic systems, which consist of flocculated particles in suspension. The consistency curve for a pseudoplastic material starts at the origin. Consequently, there is no yield value as seen in plastic systems. Moreover, since no part of the curve is linear, the viscosity of a pseudoplastic material cann’t be represented by a single value.

The viscosity of a pseudoplastic substance decreases with increasing rate of shear. An apparent viscosity can be determined at any rate of shear from the slope of the tangent to the curve at that specific point. However, the most satisfactory representation for a pseudoplastic material is likely a graphic plot of the entire consistency curve. The curved rheogram for pseudoplastic materials arises from the shearing action on long-chain molecules, such as linear polymers. As the shearing stress increases, the normally disarranged molecules begin to align their long axes in the direction of flow. This alignment reduces the internal resistance of the material, allowing for a greater rate of shear at each successive shearing stress. Additionally, some of the solvent associated with the molecules may be released, effectively lowering both the concentration and the size of the dispersed molecules, further decreasing the apparent viscosity.
The Equation of Pseudoplastic System
Newtonian systems can be fully described by their viscosity, η, and plastic systems can be adequately characterized by their yield value, f, and plastic viscosity, U. However, several approaches have been developed to obtain meaningful parameters for comparing different pseudoplastic materials. the exponential formula:
$$ F^N=\eta’G $$
has been used most frequently for pseudoplastics. The exponent N rises as flow becomes increasingly non-Newtonian. The term η’ is a viscosity coefficient. Following rearrangement, we can write in the logarithmic form:
$$ \log(G)=N\;\log(F)-\log(\eta’) $$
This is an equation for a straight line. Many pseudoplastic systems fit this equation when log G is plotted as a function of log F.
Dilatant Flow
Certain suspensions with a high percentage of dispersed solids exhibit increased resistance to flow with increasing shear rates. Such systems, which actually increase in volume when sheared, are termed dilatant; their flow properties are illustrated in Figure 3. This type of flow is the inverse of pseudoplastic flow. While pseudoplastic materials are often called “shear-thinning systems,” dilatant materials are referred to as “shear-thickening systems.” When the stress is removed, a dilatant system returns to its original state of fluidity.

Particulate systems of this type that are flocculated would be expected to exhibit plastic rather than dilatant flow characteristics. Dilatant behavior can be explained as follows: At rest, particles are closely packed with minimal interparticle volume (voids). The amount of vehicle in the suspension is sufficient to fill these voids, allowing particles to move relative to each other at low shear rates. Therefore, a dilatant suspension can be poured from a bottle because it remains reasonably fluid under these conditions. As shear stress increases, the system expands or dilates, hence the term “dilatant.”
When particles attempt to move quickly past each other, they adopt an open form of packing, as shown in Figure 3. This arrangement leads to a significant increase in interparticle void volume. Since the amount of vehicle remains constant, it eventually becomes insufficient to fill the increased voids between particles. Consequently, resistance to flow increases because the particles are no longer fully wetted or lubricated by the vehicle. Ultimately, the suspension sets up as a firm paste.
The Equation of Dilatant system
$$ F^N=\eta’G $$
This equation can be used to describe dilatancy in quantitative terms. In this case, \( N \) is always less than 1 and decreases as the degree of dilatancy increases. As \( N \) approaches 1, the system exhibits increasingly Newtonian behavior. Substances with dilatant flow properties are invariably suspensions containing a high concentration (about 50% or greater) of small, deflocculated particles.
Reference:
- Sinko, P. (2011). Martin’s Physical Pharmacy and Pharmaceutical Sciences. Baltimore, : Lippincott Williams & Wilkins, a Wolters Kluwer business.
- Felton. L. (2013). Remington Essentials of Pharmaceutics. London. UK: Pharmaceutical Press.